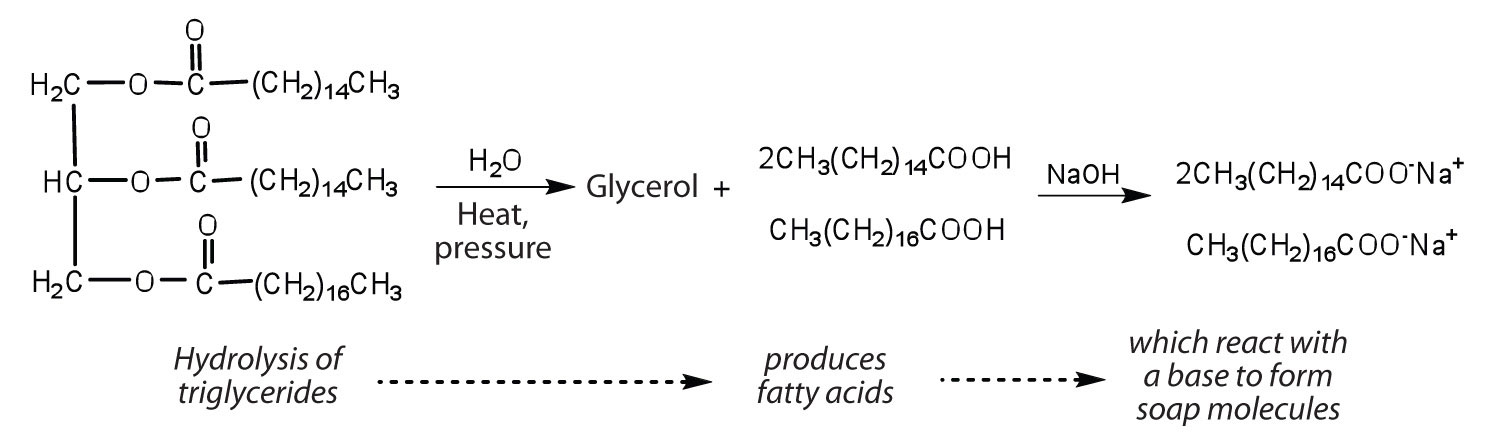
SOLVED:Soaps are produced by the reaction of sodium hydroxide with naturally occurring fats. The equation for one such reaction is C3 H5(C1 H35COO)3+3 NaOH →C3 H5(OH)3+ 3 C17 H35 COONa.The last compound
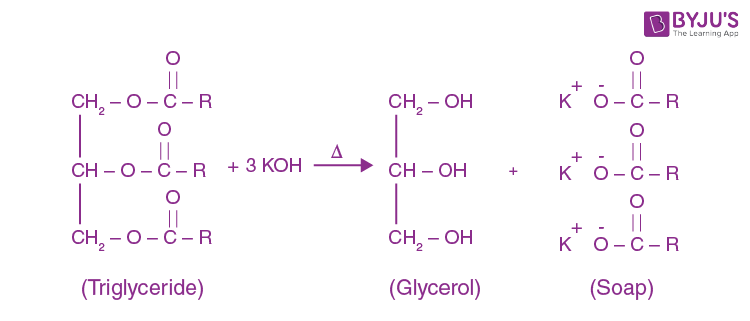
Saponification - Definition,Reactions, Mechanism, Examples, Saponification Value, Uses, FAQs with Videos on Saponification.
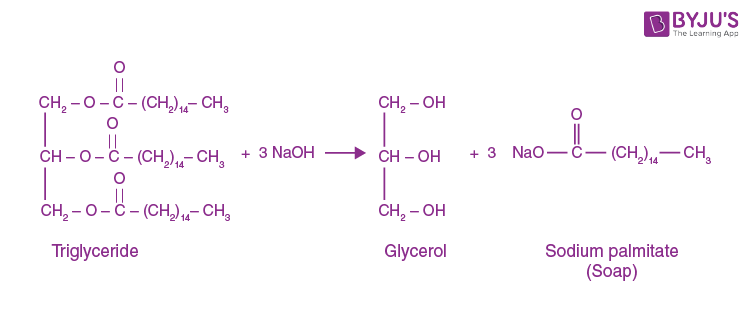
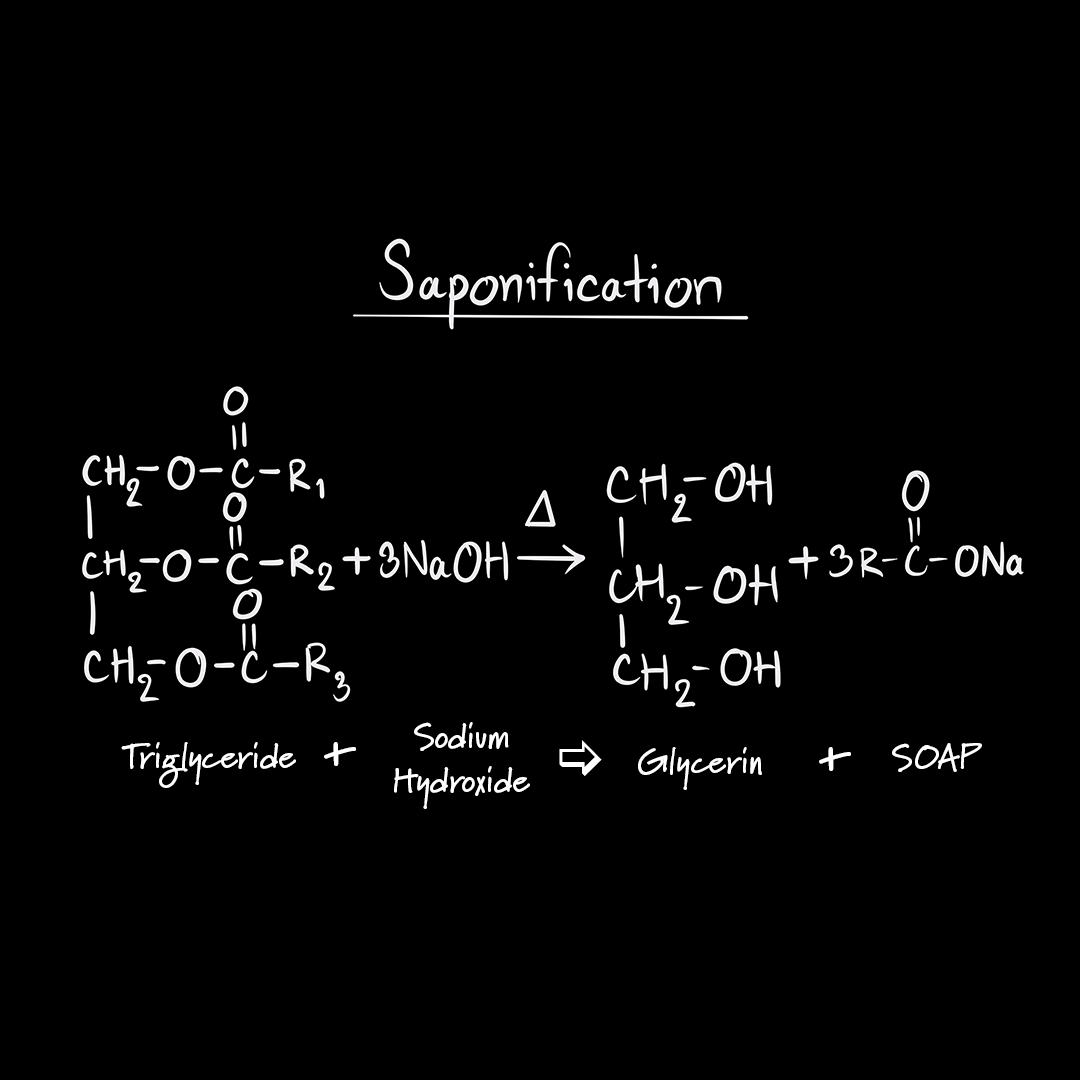


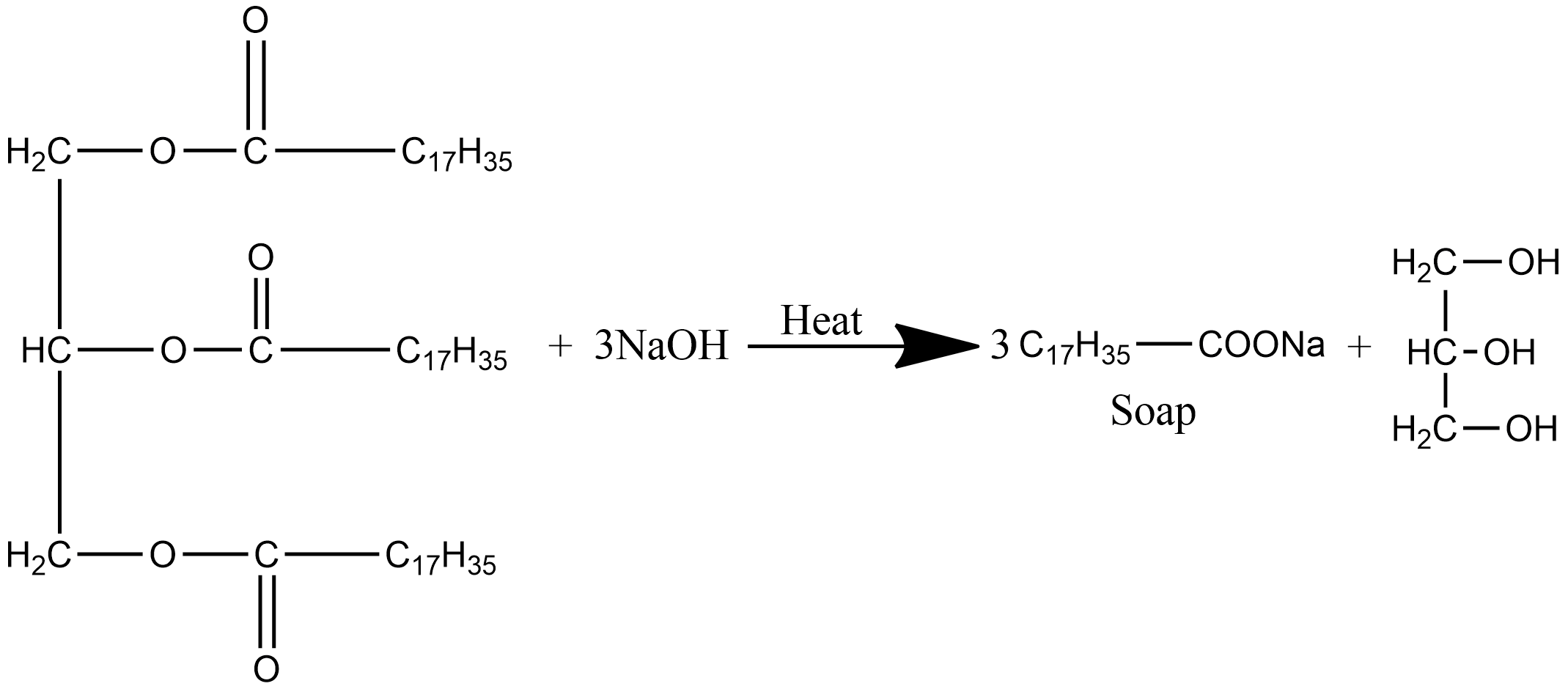
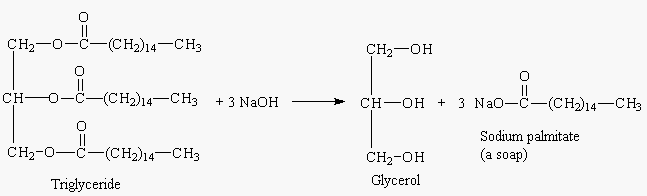


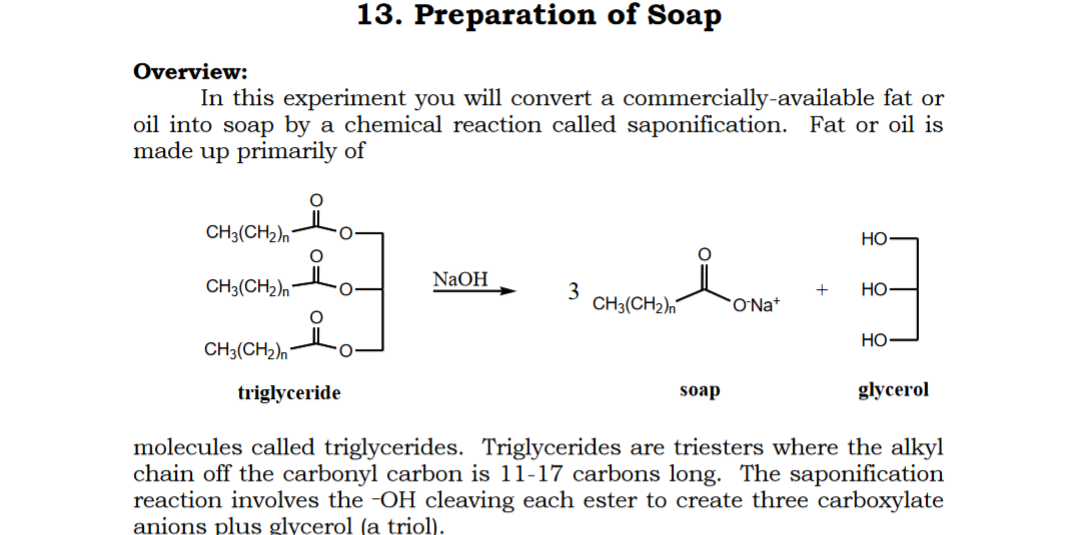
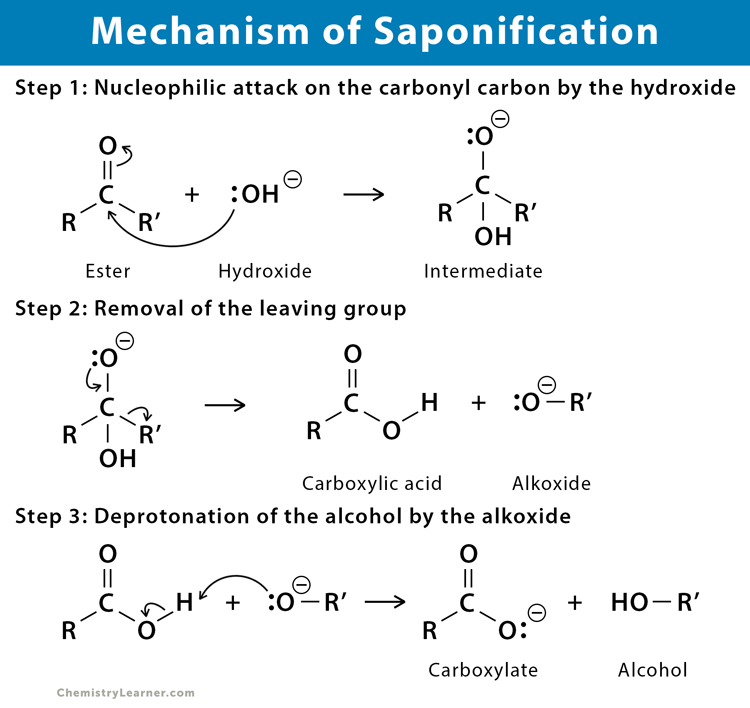

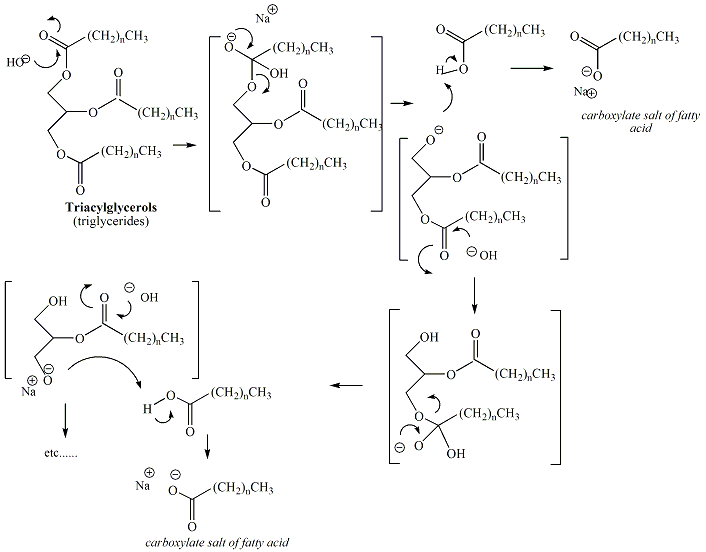

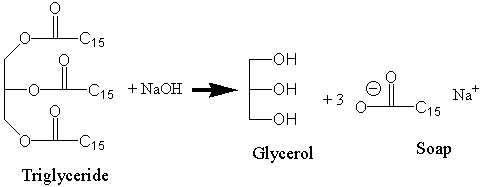
:max_bytes(150000):strip_icc()/Saponification-56a132ca5f9b58b7d0bcf749.png)