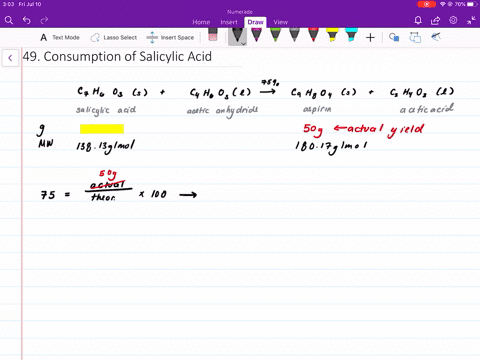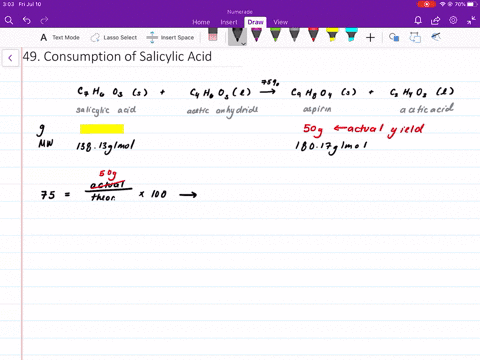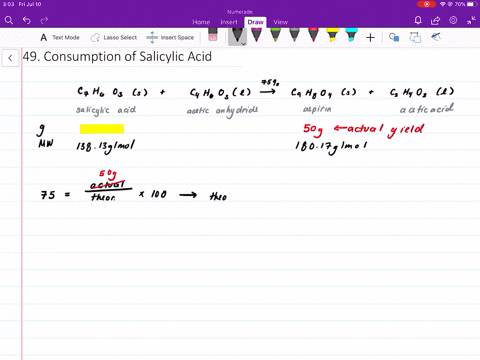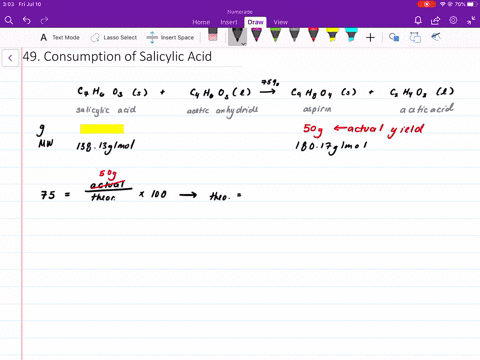
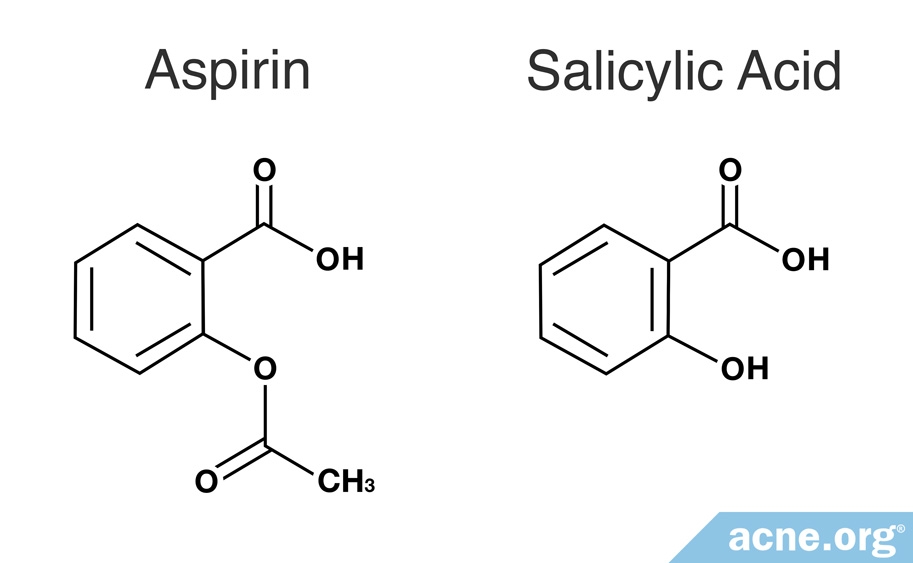
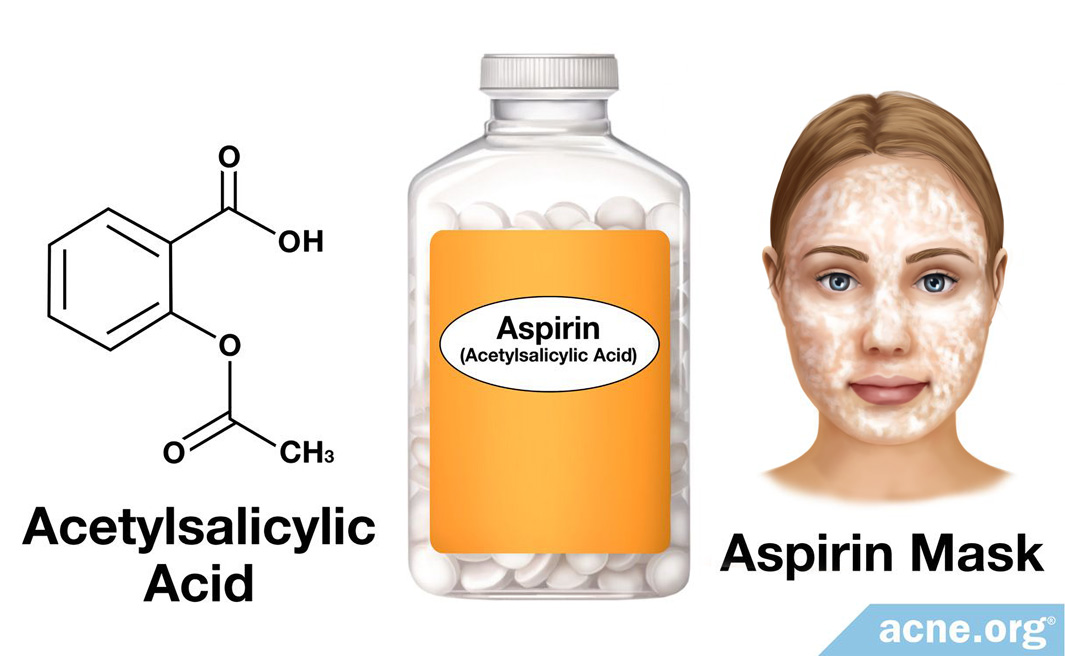
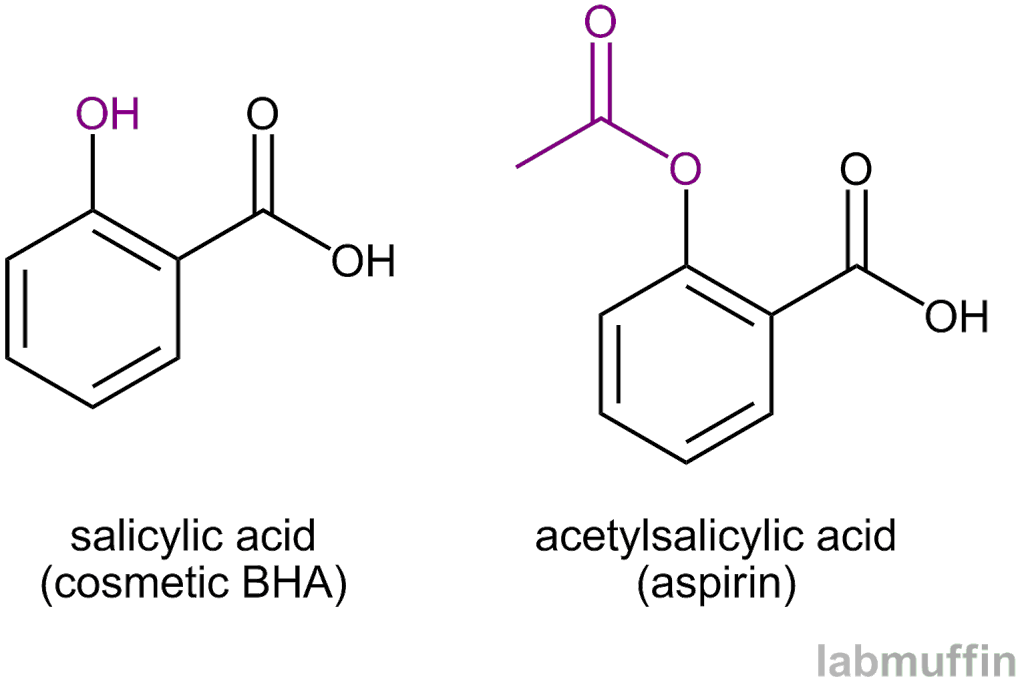
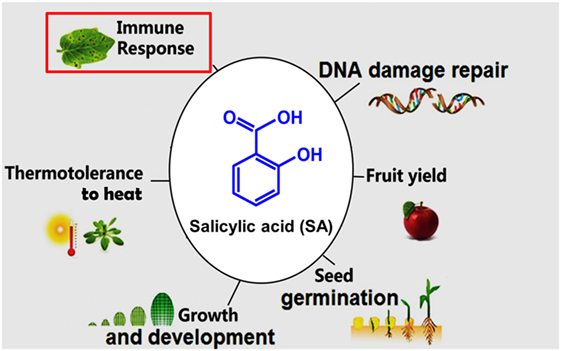
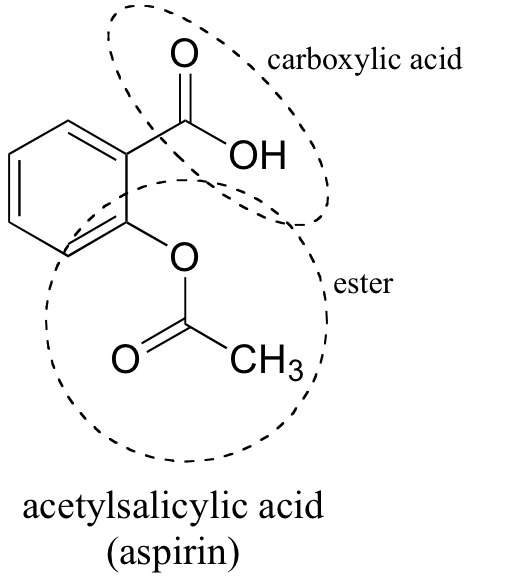
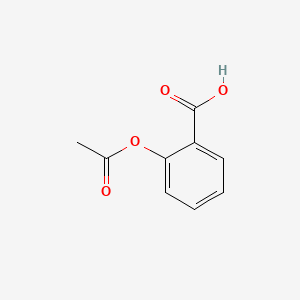
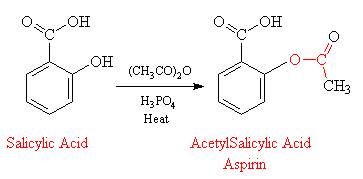
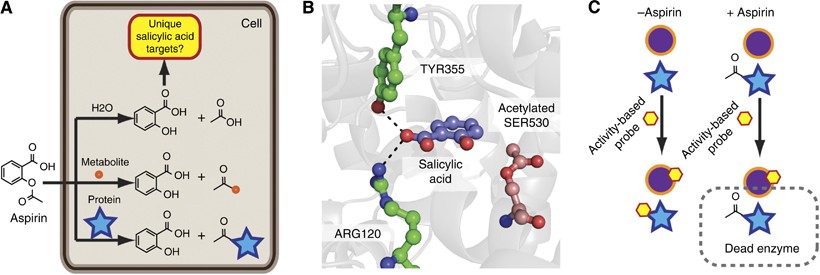
Structural representation of ASP (aspirin), SAL (salicylic acid) and IS... | Download Scientific Diagram
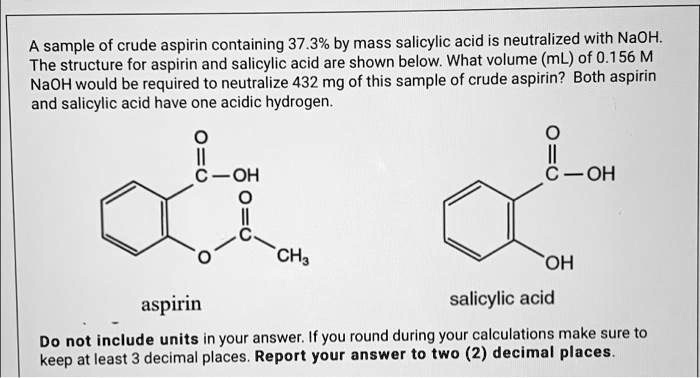
SOLVED: A sample of crude aspirin containing 37.3% by mass salicylic acid is neutralized with NaOH. The structures for aspirin and salicylic acid are shown below. What volume (mL) of 0.156 M

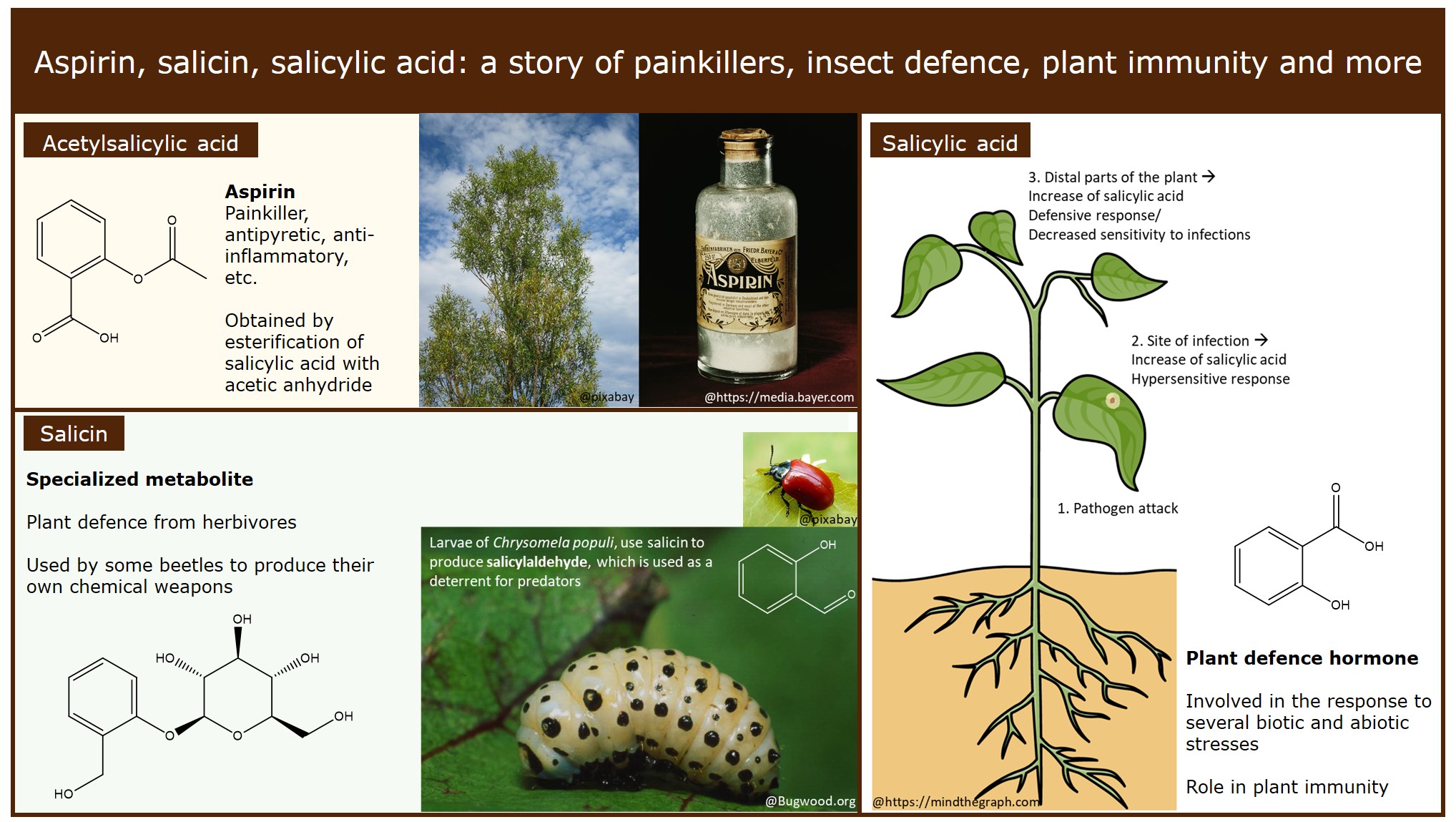
Chemical structures of salicylic acid and its synthetic and natural... | Download Scientific Diagram
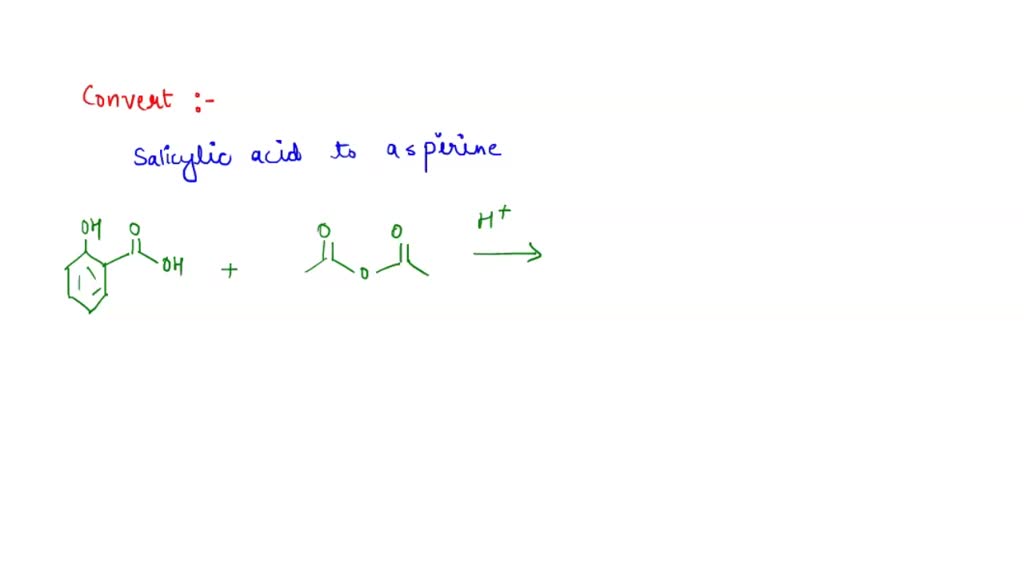
SOLVED: Carboxylic acid and phenol. Why would salicylic acid contain two acidic groups? Is it desirable to convert salicylic acid into aspirin (acetylsalicylic acid)?
![The active ingredient in aspirin is acetyl salicylic acid with\\[{{K}_{a}}=\\text{ }4.0\\times {{10}^{-9}}\\] . The pH of the solution obtained by dissolving two aspirin tablets (contain 0.36 g of acetyl salicylic acid in The active ingredient in aspirin is acetyl salicylic acid with\\[{{K}_{a}}=\\text{ }4.0\\times {{10}^{-9}}\\] . The pH of the solution obtained by dissolving two aspirin tablets (contain 0.36 g of acetyl salicylic acid in](https://www.vedantu.com/question-sets/214d4d7b-aa1e-4b43-8585-31cd4abd95614088496745223917951.png)
The active ingredient in aspirin is acetyl salicylic acid with\\[{{K}_{a}}=\\text{ }4.0\\times {{10}^{-9}}\\] . The pH of the solution obtained by dissolving two aspirin tablets (contain 0.36 g of acetyl salicylic acid in